Our Development – Milestones That Make an Impact
2020 FOUNDING
Founding of MedCare Solutions GmbH
MedCare Solutions was founded in 2020 with the goal of providing innovative medical technology that meets the highest quality standards.

2021 In-house Manufacturing
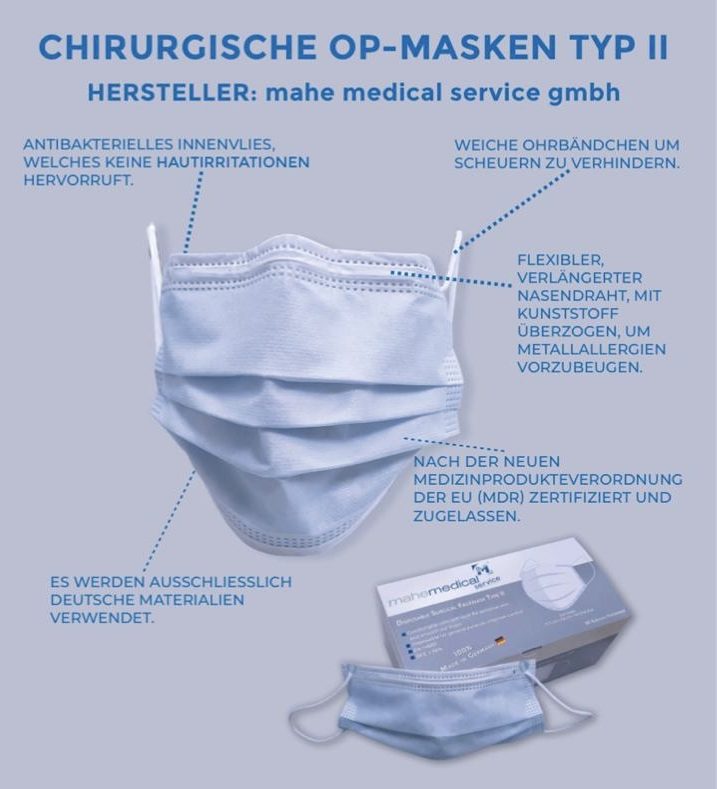
In-House Production of Type II Surgical Masks
Our Type II surgical masks are manufactured in-house according to the highest quality standards. They comply with EN 14683 and provide reliable protection as well as a high level of wearing comfort.
Thanks to modern production processes and strict quality controls, we ensure consistent quality and fast delivery times.
2021 First Certification
First Certification according to ISO 13485, as well as for Type II surgical masks and the Class IIb pedicle screw system
With the successful initial certification according to ISO 13485, as well as the certification of our Type II surgical masks and our Class IIb pedicle screw system, we meet the highest international quality and safety standards in medical technology.

2021 Establishment of Multiple Testing Centers in Baden-Württemberg
Establishment of Multiple Testing Centers in Baden-Württemberg
We successfully established several testing centers across Baden-Württemberg, including sites in Engen, Emmingen, Immendingen, Villingen, Sulz am Neckar, and Empfingen, along with additional mobile locations.

2022
Vetcare Orthopaedic – New brand
With “Vetcare Orthopaedic,” a new brand was introduced, specializing in high-quality veterinary implants.

2023
Development of TPLO Plates
Our product range was expanded to include specialized TPLO plates – developed in close collaboration with experts.

2023
Expansion of Our OEM Capabilities in the OEM Sector
With the expansion of our service portfolio, we now provide customized OEM manufacturing solutions tailored to individual customer requirements.

2024
Relocation to a New Company Building
With the introduction of our new company name and the relocation to a modern facility, we have created optimal conditions for innovation, production, and sustainable growth. The new site provides more space for development, efficiency, and long-term stability.

2024
Expiration of MDD Certification and Continuation under ISO 13485
After the expiration of our MDD certification, we seamlessly transitioned our processes to ISO 13485, continuing to ensure the highest quality standards.

2025
FDA Registration (U.S. Food and Drug Administration)
With our successful registration with the U.S. Food and Drug Administration (FDA), we meet the requirements for international market access and strengthen our presence in the U.S. market.

